Show summary Hide summary
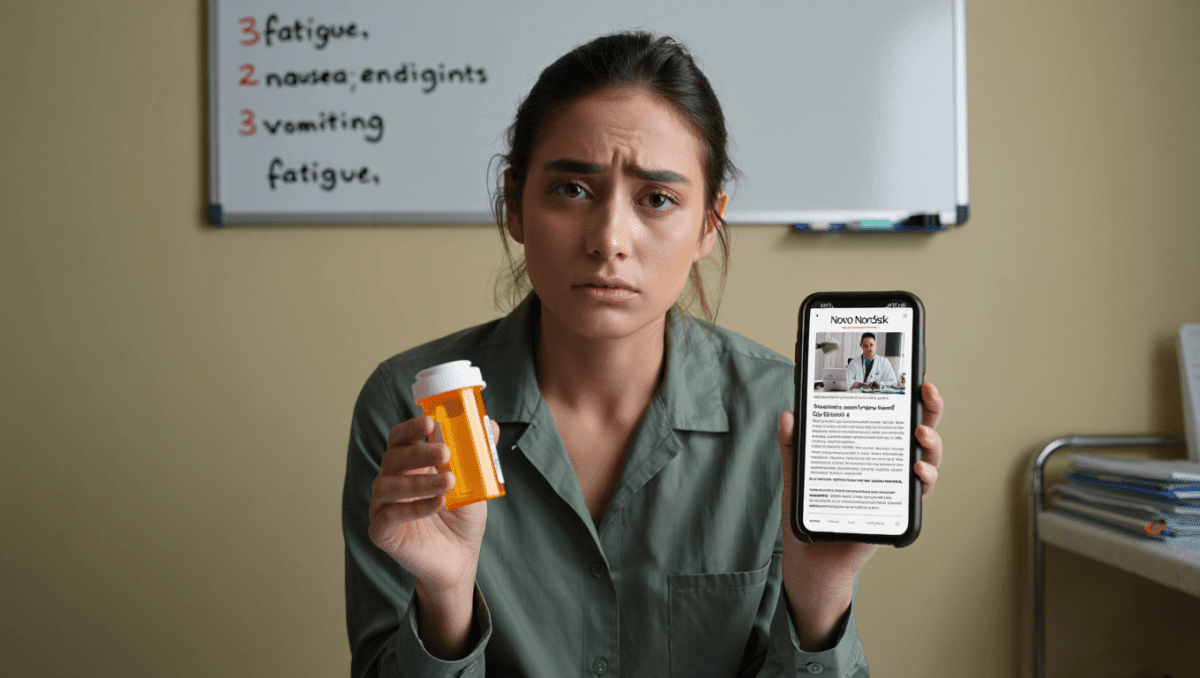
The U.S. Food and Drug Administration has formally warned drugmaker Novo Nordisk after identifying serious safety reports tied to its weight‑loss products that were not submitted to regulators within required timeframes. The agency’s notice specifically referenced two deaths and a report of suicidal ideation among the cases it said were reported late — a development that raises fresh questions about post‑market monitoring as use of GLP‑1 medicines grows rapidly.
What the FDA flagged
The FDA’s communication to the company centers on delayed notification of incidents the agency classifies as serious adverse events. Timely reporting of such events is a core part of how the agency tracks emerging safety risks after a drug reaches the market.
Rigetti stock surges 20% on government quantum investment, gains 50% this week
Saving money: High-yield accounts hit 5.00% APY as US rates climb
- Two deaths were cited among the cases not submitted within the required reporting windows.
- One report involved suicidal ideation that, according to the FDA, was not timely communicated.
- The agency raised concerns about the company’s overall reporting practices for other post‑market events.
Regulators do not disclose full details of every event in a warning, so the public record often leaves unanswered questions about timing, circumstances and the specific products involved. The FDA’s letter, however, signals the agency found the lapses serious enough to merit formal notice.
Why this matters now
The timing is important because prescriptions for GLP‑1 therapies such as Wegovy and Ozempic have surged in recent years, expanding their user base beyond patients with diabetes to include people seeking weight loss. That expansion makes rapid, accurate adverse‑event reporting essential: delayed signals can slow safety investigations, label changes or other measures that protect patients.
For clinicians and patients, the practical risk is clear — if safety data are slow to reach regulators, decisions about prescribing and monitoring are made with incomplete information. For the company, late reporting can prompt regulatory follow‑up that ranges from letters and inspections to potential enforcement actions.
Possible outcomes and next steps
- Regulatory follow‑up: The FDA may request corrective plans, conduct inspections or seek additional data to assess the scope of reporting failures.
- Label and product reviews: If patterns emerge, the agency could require updates to safety warnings or patient materials.
- Industry scrutiny: Other regulators and healthcare organizations may increase surveillance of similar drugs and manufacturers.
Patients currently using these medications should not make abrupt changes based on the warning alone. Instead, experts say patients worried about side effects should talk to their prescribing clinician to review symptoms and individual risk. Health care providers must remain alert to new safety signals and report suspected serious events promptly.
When reached for comment, Novo Nordisk did not immediately respond to requests from reporters. The FDA’s action underscores broader pressure on regulators and manufacturers to keep pace with a fast‑growing market for weight‑loss therapies and to ensure that safety monitoring remains timely and transparent.