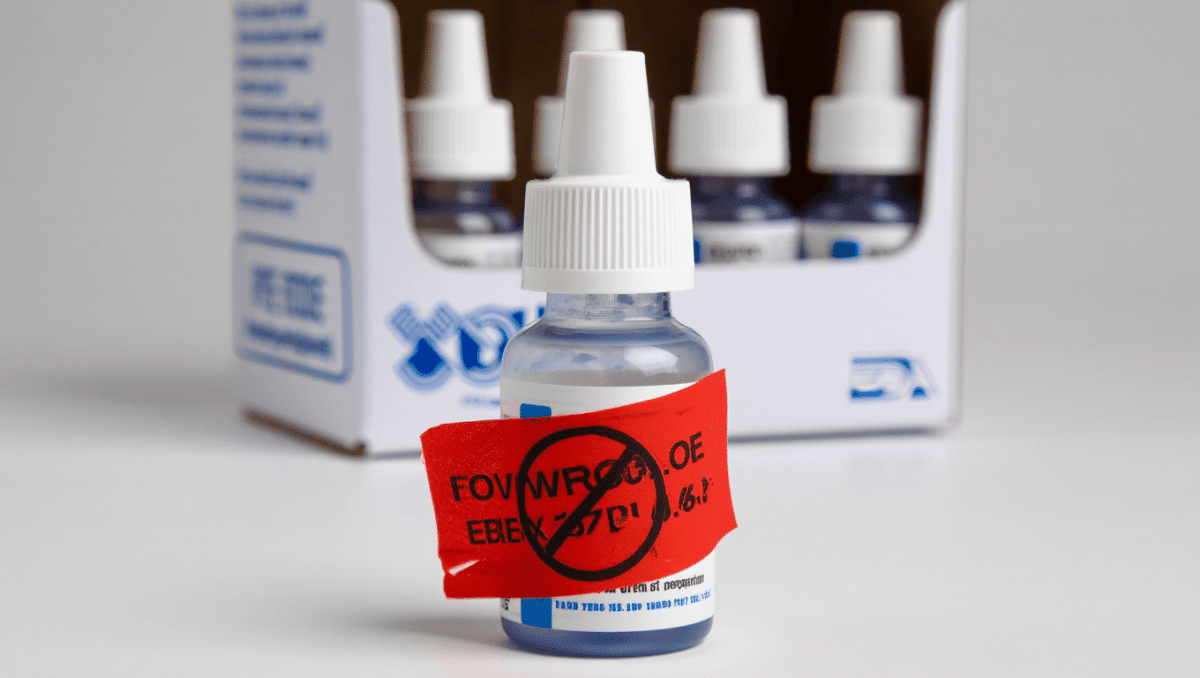
The U.S. Food and Drug Administration has warned consumers about a broad recall affecting about 3.1 million store-brand eye drops, saying the manufacturer cannot confirm the products are free from bacterial or fungal contamination. No injuries have been reported so far, but regulators say the potential for harmful organisms in these over-the-counter solutions makes the recall urgent.
The recall applies to a large batch of retailer-branded lubricating eye drops distributed nationwide, the FDA said in its notice. While specific product names and lot numbers were not included in the initial announcement, the agency emphasized that the issue centers on compromised sterility — a primary safety requirement for liquid ophthalmic products.
What sterility failures mean for users
Eye drops are designed to be sterile to avoid introducing bacteria or fungi to the eye, where even minor contamination can lead to serious infections. The FDA’s action reflects the risk that contaminated solutions could cause inflammation, corneal ulcers or other complications that sometimes require antibiotics or specialist care.
Regulators typically classify such recalls as precautionary when contamination is suspected but no adverse events have yet been confirmed. That distinction matters for consumers who may not feel immediate effects but could still face delayed symptoms.
Immediate steps consumers should take
- Stop using any store-brand eye drops you suspect may be part of the recall.
- Check the packaging and receipts for retailer identifiers and lot numbers, and compare them to any recall list posted by the FDA or the retailer.
- Return the product to the place of purchase or contact the retailer or manufacturer for a refund or replacement.
- Do not attempt to reuse or transfer remaining liquid to other containers.
- If you have used the drops and notice eye pain, worsening redness, discharge, light sensitivity, or changes in vision, seek medical attention promptly.
Signs of a possible eye infection
Not all irritation comes from contamination, but several symptoms warrant quick evaluation: persistent redness beyond normal irritation, increasing pain, a sticky or colored discharge, or sudden vision changes. Infections of the cornea can progress rapidly and may require prescription treatment to prevent lasting damage.
Primary care clinicians, urgent care centers, or ophthalmologists can assess symptoms and prescribe appropriate therapy. If symptoms are severe — such as intense pain or major vision loss — patients should seek emergency care.
Wider implications
Even without reported injuries, recalls of this scale raise questions about manufacturing controls and quality assurance for store-brand products, which are often produced by third-party manufacturers. The FDA may follow up with inspections or require corrective actions to prevent future lapses.
For now, the key takeaway is practical: treat the recall seriously, check your medicine cabinet, and err on the side of caution. Retailers typically post recall details and refund instructions on their websites; consumers should consult the FDA recall page for updates and for any lists that identify specific products and lot numbers.
Bottom line: If you have recently purchased store-brand lubricating eye drops, stop using them until you can confirm the product is unaffected by the recall. Monitor your eyes for symptoms and contact a health professional if you have any concerns.